Medical Writing Market Growth, Segments & Forecast 2025–2033
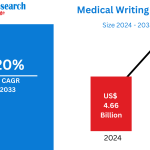
According to Renub Research global medical writing market is experiencing strong and sustained growth, driven by the expanding pharmaceutical and biotechnology industries and the rising complexity of regulatory requirements. The market is projected to grow from US$ 4.66 billion in 2024 to US$ 12.12 billion by 2033, registering a robust CAGR of 11.20% from 2025 to 2033.
Key factors fueling this growth include the increasing number of clinical trials, heightened focus on drug safety and compliance, growing demand for regulatory documentation, advancements in digital and AI-enabled writing tools, rising trends toward outsourcing, and the expansion of personalized medicine and health communication initiatives.
Medical Writing Market Overview
Medical writing refers to the preparation of scientifically accurate, clear, and regulatory-compliant documents related to healthcare, medicine, and drug development. These documents include clinical study reports, regulatory submissions, scientific manuscripts, educational materials, and medical marketing content. Medical writers serve as a critical bridge between scientific data and its effective communication to regulatory authorities, healthcare professionals, and the general public.
As healthcare research becomes increasingly complex and globalized, medical writing has become an essential function within the pharmaceutical, biotechnology, and medical device industries. Writers must possess a deep understanding of clinical research processes, medical terminology, ethical standards, and global regulatory guidelines to ensure consistency, transparency, and compliance.
Request a free sample copy of the report:https://www.renub.com/request-sample-page.php?gturl=medical-writing-market-p.php
Global Medical Writing Industry Overview
The global medical writing industry supports the entire lifecycle of drug and device development—from early-stage research documentation to post-marketing surveillance and scientific dissemination. Increasing globalization of clinical trials and stricter regulatory scrutiny have significantly elevated the demand for high-quality documentation.
Digital transformation has further reshaped the industry, with electronic submissions, online journals, and digital health platforms replacing traditional publishing models. Advances in artificial intelligence, automation, and data analytics are also improving efficiency while increasing output quality. Additionally, aging populations and expanding healthcare access in emerging markets are contributing to sustained industry growth.
Growth Drivers for the Medical Writing Market
The medical writing market is driven by multiple interrelated factors, including increased clinical research activity, regulatory complexity, and global healthcare expansion. Pharmaceutical and biotechnology companies increasingly rely on professional writing services to manage documentation workloads efficiently while maintaining compliance with evolving international regulations.
Outsourcing non-core activities such as medical writing allows organizations to reduce costs, improve turnaround times, and access specialized expertise, further accelerating market growth.
Rising Number of Clinical Trials and Medicinal Approvals
The growing volume of clinical trials and drug approvals worldwide is a primary driver of medical writing demand. Each clinical trial generates extensive documentation, including protocols, investigator brochures, clinical study reports, and regulatory submissions. As pharmaceutical pipelines expand, the need for accurate, timely, and compliant documentation increases.
In addition, regulatory agencies require comprehensive and standardized documentation to evaluate drug safety and efficacy. This has led to a rising demand for experienced medical writers capable of producing high-quality submissions across multiple therapeutic areas and regulatory jurisdictions.
Rising Research and Development Activities
Increased R&D investment by pharmaceutical, biotechnology, and medical device companies is significantly fueling the medical writing market. As research activities intensify, organizations require detailed documentation such as clinical trial protocols, statistical analysis plans, and peer-reviewed publications.
In January 2023, Cactus Communications acquired FlexSteel Technologies Holdings, Inc. in a deal valued at US$ 621 million, highlighting the growing importance of scientific communication and medical writing capabilities. This acquisition reflects the strategic value placed on expanding expertise in technical and medical content development.
Globalization of Drug Development
The globalization of drug development has dramatically increased the demand for professional medical writing services. Pharmaceutical and biotechnology companies conduct clinical trials across multiple countries, each with unique regulatory requirements. This creates a need for standardized yet localized documentation that complies with diverse regulatory frameworks.
Medical writers play a critical role in harmonizing global submissions, adapting content for different regulatory authorities, and ensuring consistency across multinational clinical programs. Language localization and cultural adaptation further enhance the demand for skilled professionals with international regulatory expertise.
Challenges in the Medical Writing Market
Despite strong growth prospects, the medical writing market faces several operational and talent-related challenges. Maintaining quality, consistency, and regulatory compliance across complex projects remains a significant concern for service providers and sponsors alike.
Maintaining Consistency and Quality
Ensuring consistency and quality is one of the most critical challenges in medical writing. Projects often involve multiple contributors, including clinicians, statisticians, regulatory experts, and reviewers, increasing the risk of inconsistencies in terminology, tone, and data interpretation.
Large, multi-author documents require rigorous quality control processes, including standardized templates, version control systems, and multiple rounds of review. Maintaining compliance with strict regulatory and ethical standards demands meticulous attention to detail, making quality assurance both time-consuming and resource-intensive.
Shortage of Skilled Medical Writers
The growing demand for medical writing services has resulted in a shortage of skilled professionals with both scientific knowledge and regulatory expertise. The specialized nature of medical writing, combined with constantly evolving regulatory requirements, makes talent acquisition challenging.
This shortage can lead to project delays, increased competition for qualified writers, and rising labor costs. To address this issue, companies are investing in training programs and leveraging technology to enhance productivity and reduce dependency on scarce talent.
United States Medical Writing Market
The United States represents the largest medical writing market globally, driven by a strong pharmaceutical and healthcare ecosystem. The high number of clinical trials, particularly in oncology, cardiology, and neurology, significantly increases demand for clinical and regulatory documentation.
The growing need for clinical study reports, regulatory filings, and scientific publications is expected to sustain market growth. The U.S. also benefits from advanced research infrastructure, regulatory expertise, and a strong presence of global medical writing service providers.
India Medical Writing Market
India’s medical writing market is expanding rapidly due to the country’s growing pharmaceutical and biotechnology industries and its position as a global hub for clinical trials. A large pool of highly skilled, English-speaking professionals makes India an attractive destination for outsourcing medical writing services.
Cost-effective service delivery, increasing adoption of AI tools, and supportive government initiatives further strengthen India’s competitive advantage. As global companies continue to seek efficient and scalable solutions, India’s role in the medical writing market is expected to grow significantly.
United Kingdom Medical Writing Market
The United Kingdom medical writing market is supported by a robust healthcare system and a well-established pharmaceutical and biotechnology sector. Rising clinical trial activity and stringent regulatory requirements drive demand for both clinical and regulatory writing services.
Clinical writing remains the largest revenue-generating segment, while regulatory writing is experiencing rapid growth. The UK’s skilled workforce and strong research environment position it as a key player in the global medical writing landscape.
United Arab Emirates Medical Writing Market
The medical writing market in the United Arab Emirates is growing steadily as the country strengthens its healthcare infrastructure and expands clinical research activities. The UAE’s focus on healthcare innovation, medical tourism, and compliance with international regulatory standards supports market expansion.
Digital health adoption and increasing demand for scientific and regulatory documentation further contribute to growth, positioning the UAE as an emerging hub for medical writing services in the Middle East.
Medical Writing Market Segmentation by Type
The medical writing market is segmented into clinical writing, regulatory writing, scientific writing, and other specialized writing services. Clinical writing accounts for the largest share due to extensive documentation requirements in clinical trials, while regulatory writing is growing rapidly in response to stricter global regulations.
Medical Writing Market Segmentation by Application
Key applications include medical journalism, medical education, medico-marketing, and other communication services. Increasing demand for evidence-based content and health communication initiatives is expanding application areas across the industry.
Medical Writing Market Segmentation by End Use
End users include contract research organizations (CROs), pharmaceutical companies, biotechnology firms, and medical device manufacturers. CROs represent a major share due to their involvement in large-scale clinical trials and regulatory submissions.
Medical Writing Market Segmentation by Geography
The market is analyzed across North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. North America leads in revenue, while Asia Pacific is the fastest-growing region due to outsourcing trends and expanding healthcare infrastructure.
Competitive Landscape of the Medical Writing Market
The medical writing market is highly competitive, with companies focusing on service expansion, strategic partnerships, and technology adoption. Leading players include Parexel International Corporation, Trilogy Writing & Consulting GmbH, Freyr, Labcorp Drug Development, IQVIA Holdings Inc., Omics International, and Synchrogenix.
Companies are evaluated based on overview, leadership, recent developments, strategic initiatives, and sales performance.
Conclusion
The global medical writing market is poised for strong growth through 2033, supported by expanding clinical research activity, increasing regulatory complexity, and rising demand for high-quality scientific communication. While challenges such as talent shortages and quality assurance persist, technological advancements and outsourcing trends are expected to sustain long-term market expansion. Medical writing will remain a critical enabler of global healthcare innovation and regulatory compliance.
RENUB